| |
|
Scroll
down to browse through some archived SWIMMING POOL
questions and answers. Please click the Pool
Problems Link, on top of every page, to access a complete
listing of Pool Problem subjects, an alphabetized
Website Table of Contents, Pool Equipment Information,
About Alan Biographic Material and a Pool Glossary. Use
the other links to access additional subject
information. More information about some new and unique
products, for pools and spas, can be found by visiting
The Website Store.
You'll never know what you'll find and that's always
fun. Be better prepared and avoid costly problems!
|
|
 |
Shipping is FREE* . . . within
the Continental U.S.
A $9.99
handling charge will apply
to Continental U.S. Orders,
under $75.00. Orders
outside of the Continental
U.S. may require some
additional charge, based on
quantity and destination.
Most products can be
shipped World-Wide. International
and orders outside of
Continental U.S. - see
comments on the ordering
pages. |
|
 |

Major
Credit Cards and PayPal are accepted. |
|
On-line
shopping since 2002 - Safe and Secure!!! |
|
Managing the Chlorine level, of pool
water!!! |
|
Commercial pools are expected to
be maintained to a higher
standard, than residential
pools. High bather usage
places extra demands on
sanitizing, water chemistry,
filtration and safety. A
ColorQ, all digital Water Tester
can perform all of the common
tests, while eliminating the
color-matching and guesswork.
With 10 models, performing up to
11 different test factors, one
is right for every need.
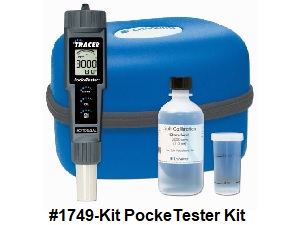
An
Electronic
PockeTester Kit is a convenient
way to monitor the salt level. The
WaterLink SpinTouch Labs
are the ultimate tester, doing
up to 10 different water test
factors, in just 1 minute.
Maintains a
past history and allows for the
remote monitoring of test
results. Voted product of
the year, by the trade
association. |
|
ColorQ All-Digital Water Testers |
Testing The Salt Level |
WaterLink SpinTouch Labs |
 |
 |
 |
|
Product and Ordering Information |
Product and Ordering Information |
Product and Ordering Information |
If you have a pool or spa water
testing need, we should have the
product.
►
Scroll down to read through some
Question & Answer information.
◄ |
|
|
A
structural, concrete crack requires more
than just epoxy.
Torque-Lock
Staples use 5000 pounds of
torque, to hold the opposite sides
together and make a rock solid repair.
Ultra Poly One Coat is a
hybrid-epoxy coating, that is easier to
prep and apply and can be used to
refinish masonry or fiberglass pools,
spas and fountains. Used in
commercial pools, water parks and water
features, all across the U.S.
Keep the area around a pool or spa
cleaner and they stay cleaner. A
MetalTrap
Dual-Cartridge Filter attaches
to a garden hose and can be used to
remove sediments and dissolved copper,
iron and manganese, from new water
additions. |
|
|
|
How to
manage a commercial swimming pool? |
Commercial
Pools cannot always use the same chemicals that
are used in typical residential pools, because
of the regulations of local health departments.
What is permitted will vary from state to state.
High bather usage can require specialized
equipment and sanitizing practices. Make sure
that your pool is in compliance with the latest
anti-entrapment requirements. If problems arise,
refer to the
Pool Problems Page, as a source of
problem-solving information, broken down into
various categories. Scroll down the page
and click on the linked
keywords,
catch phrases
or images, in the archived answers below, to access additional information, on that topic or product.
 |
Join our E-Letter Mailing List.
You'll receive 1-3 E-Letters a
month, featuring helpful pool
and spa advice, new product
information and sale
announcements. All we
require is your e-mail address
and you can opt out anytime you
wish.
Your information
will never be shared or sold. |
|
▼
Helpful,
Problem-Solving Information, in a question and
answer format.
▼ |
► Commercial Pool
Chlorination?
I recently started taking care of a
commercial pool and am having problems getting the chlorine
levels up. It seems that no matter how many times I
clean/replace the filters, add trichlor sticks or shock with
lithium, I can't get the chlorine level any higher than 1 on
my 4-way test strips. Any suggestions. Thanks in Advance.
Rick S., Bolton Valley, VT,
4/23/2019
There are basically two possibilities here. The chlorine
requirements could be so high that the chlorine sticks
cannot keep up with the demand. Unless the chlorine feeder
is oversized and full, it may not be able to dissolve the
product at a fast enough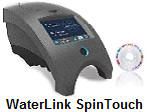 rate and that explains the need for
the shock treatment. Trichlor is not the best way to
sanitize a commercial pool. The other possibility is that
the test strips are not reading properly. This can happen,
if the strips are removed with wet fingers or not stored
properly. I suggest that you use something else to verify
the results. In either case, you must use products and
testers that are allowed for use in commercial pools. State
laws vary, on these matters. A
WaterLink SpinTouch Lab is not
only the fastest way to do a
comprehensive water analysis, it is
easy to use. It keeps track of
the previous tests results and all
testing can be monitored, from a
remote location, using cloud-based
software and a computer. Even
multiple locations can be centrally
monitored, so long as each is using
a WaterLink SpinTouch Lab. When the
pool operators know someone can be
watching, they are more likely to
stay on top of things. Has anyone ever
thought about upgrading the sanitizing? A
salt chlorine
generator would be an excellent choice, as it is a benefit
to both the operator and the pool users. Another thing to
consider is the use of ultraviolet sanitizing. It won't
replace the chlorine, but will greatly reduce the amount of
chlorine required, dramatically reduce the chloramine odor
and simplify the water chemistry. I hope that this
information will prove helpful. rate and that explains the need for
the shock treatment. Trichlor is not the best way to
sanitize a commercial pool. The other possibility is that
the test strips are not reading properly. This can happen,
if the strips are removed with wet fingers or not stored
properly. I suggest that you use something else to verify
the results. In either case, you must use products and
testers that are allowed for use in commercial pools. State
laws vary, on these matters. A
WaterLink SpinTouch Lab is not
only the fastest way to do a
comprehensive water analysis, it is
easy to use. It keeps track of
the previous tests results and all
testing can be monitored, from a
remote location, using cloud-based
software and a computer. Even
multiple locations can be centrally
monitored, so long as each is using
a WaterLink SpinTouch Lab. When the
pool operators know someone can be
watching, they are more likely to
stay on top of things. Has anyone ever
thought about upgrading the sanitizing? A
salt chlorine
generator would be an excellent choice, as it is a benefit
to both the operator and the pool users. Another thing to
consider is the use of ultraviolet sanitizing. It won't
replace the chlorine, but will greatly reduce the amount of
chlorine required, dramatically reduce the chloramine odor
and simplify the water chemistry. I hope that this
information will prove helpful.
Sincerely. Alan Schuster, 4/23/2019
► Controlling
The Water Chemistry?
We run a small community pool and
would like to research a better means of adding chemicals.
The pool uses liquid chlorine and acid and these are added
with feeding pumps, tied into the operation of the filter
and pump. The problem is that the bather use is anything but
consistent and sometimes there is too much chlorine and
other times there is not enough. Are there controllers that
can be added? How expensive are they? Thanks for the help.
B. M., Knoxville, TN, 6/23/2020
Yes, controllers can be added that will help regulate the
addition of the chlorine and acid. It should give you more
uniform conditio ns. So far as the cost of these controllers
is concerned, I will have to pass. There are several
manufacturers of such equipment. I suggest that you consult
with a pool company that deals with non-residential pools.
There are other chemical saving and highly controllable
sanitizing methods, that are suitable for use in
commercial-type pools. Ozonation, for example, would deal
with the introduction of bather wastes and would decrease
the amount of chlorine required to maintain a satisfactory
level. Salt chlorine generators and
UV Sterilizers are other
possibilities. Browse through the pages on these topics for
more information. Sorry, that I couldn't provide all of the
details. ns. So far as the cost of these controllers
is concerned, I will have to pass. There are several
manufacturers of such equipment. I suggest that you consult
with a pool company that deals with non-residential pools.
There are other chemical saving and highly controllable
sanitizing methods, that are suitable for use in
commercial-type pools. Ozonation, for example, would deal
with the introduction of bather wastes and would decrease
the amount of chlorine required to maintain a satisfactory
level. Salt chlorine generators and
UV Sterilizers are other
possibilities. Browse through the pages on these topics for
more information. Sorry, that I couldn't provide all of the
details.
Sincerely. Alan Schuster, 6/24/2020
► Reducing
The Chlorine Odor?
I operate a commercial indoor pool
that gets a lot of use. The odor is a real problem with some
of the users. I know that chloramines are the cause, but I
can't keep on adding shock, as the pool is very high volume.
I have heard the ultraviolet can make a difference. Is there
any truth to that? Thanks for the time spent.
Frank B., 5/18/2013
In indoor high-use commercial pools, odor can be an
unpleasant feature. You will find the environment of the
pool to be vastly improved, within 1-3 days, after
installing an
Ultraviolet Sanitizer System. The UV rays not
only sanitize the water, but dramatically reduce or
eliminate the chloramines. When chlorine combines with
nitrogen-ammonia compounds, chloramines are formed and
produce the chlorine smell that is so obnoxious, irritating
and unhealthy. Less chlorine will be required to maintain
the customary levels and the overall water chemistry will be
less subject to fluctuation and buildup problems. I hope
that I have explained some of the benefits.
Sincerely. Alan Schuster, 5/18/2013
► Chloramines
And Ionization?
A public pool, that I swim at has had
chronic chloramine build up that has resulted in air quality
issues. The pool is approximately 125,000 gallons with a
high bather load and uses liquid chlorine and muriatic acid.
I know there are a lot of factors that go into reducing
chloramines, but my question is specific to a copper/silver
ionizer. This equipment was installed at this pool for the
sole purpose of reducing chloramines, but there has been no
real improvement. According to the owner, it is working, but
you wouldn’t know it. Chloramines are consistently above 1
ppm and the air is heavy with the odor. They also state that
they shock the pool with liquid chlorine to reach
breakpoint. They do not use MPS for shock treatment. My
question is whether or not the ionization system, they have
in place, is actually capable of breaking down chloramines?
My understanding of copper/silver is that they are
sanitizers via ionization, but not strong oxidizers. Can you
tell me if there are merits to this system with respect to
chloramine reduction? Thanks for any insight. You provide a
great service.
Richard W.,1/26/2008
You seem to have an excellent understanding of the issues.
Ionization alone will not eliminate or reduce the chloramine
concentra tion. It should reduce the amount of chlorine
required to maintain a proper free chlorine level, but will
have no effect on chloramines. To destroy or reduce the
chloramine content, oxidation is required. It can be in the
form of chlorine or potassium monopersulfate.
Ozone Generators will reduce chloramines and
odors very effectively, but will still need some chlorine to
act as a backup sanitizer. A salt chlorine generator would
have been a better choice, as it manufactures chlorine,
right in the pool. The water would be stripped of
chloramines, as water passes through the salt cell. State
law might enter into which equipment is best suited for a
public or commercial pool and this should be considered. I
think a better choice could have been made. I hope that this
information will prove helpful. tion. It should reduce the amount of chlorine
required to maintain a proper free chlorine level, but will
have no effect on chloramines. To destroy or reduce the
chloramine content, oxidation is required. It can be in the
form of chlorine or potassium monopersulfate.
Ozone Generators will reduce chloramines and
odors very effectively, but will still need some chlorine to
act as a backup sanitizer. A salt chlorine generator would
have been a better choice, as it manufactures chlorine,
right in the pool. The water would be stripped of
chloramines, as water passes through the salt cell. State
law might enter into which equipment is best suited for a
public or commercial pool and this should be considered. I
think a better choice could have been made. I hope that this
information will prove helpful.
Sincerely. Alan Schuster, 1/26/2008
► Clouded Up?
I am on the board of a homeowners pool
and a recent grad of a CPO course however, I need some help
on a problem. Our pool is 80,000 gallons. The pH reading is
7.2, FAC is 2 ppm, TA is 220. We use Calcium Hypochlorite as
our sanitizer. Our pool opened today and the water was
crystal clear. The ambient temperature was 91 °F. Not sure
of water temperature. Four hours into our first day the
water started to become cloudy. Two hours later very cloudy.
We backwashed the filters. Additionally, according to my CPO
book we should try the acid column to convert the C03 to
C02. I have read several of your answers regarding TA. I am
more concerned with the cloudiness. Any help?
Doug, 5/18/2010
It would have been nice to know what the calcium hardness
reading was? The use of calcium hypochlorite will raise the
calcium level, the pH and the TA. Your TA is already too
high and it is likely that the calcium reading is in the
hundreds of PPM. That can account for the cloudy pool water
problem. Depending on the makeup of the dissolved mineral,
lowering the TA might prove difficult, but it should be
tried. I suggest that you consider using something other
than calcium hypochlorite. Liquid chlorine would seem
logical. Even better would be a
salt chlorine generator.
Otherwise, the increasing calcium hardness levels will only
cause more and more pool water clarity problems. Adding
mineral treatments will help to an extent, but is not a long
term solution. I hope that I have been of assistance. Good
luck.
Sincerely. Alan Schuster, 5/19/2010
► ORP
Confusion?
Having a bit of a bizarre issue with
the pool which I run! The Alkalinity is always a bit low
(65-75ppm) so have been adding sodium bicarbonate to the
pool to bring it up a bit, I use sodium hypochlorite as the
disinfectant. The levels of disinfectant are measured in
redox only (this is the only one I've ever come across that
does not have the PPM reading on as well) which only
measures the disinfectant activity and not quantity. Every
time, I add sodium bicarbonate the chlorine goes up. does
adding bicarbonate interfere with the redox reading, thus
causing it to dose more chlorine? Or is it just a freaky
coincidence? I really hope you can help as most people run
at the mention of redox controllers. Not sure buying a new
controller is an option? Yours in anticipation.
Julie, United Kingdom, 8/15/2005
It is not a matter of interference. The controller is
measuring the Oxidation-Reduction Potential (ORP). As
expected, adding c hlorine causes the reading to rise. The
ORP is based on the hypochlorous acid concentration, which
rises as the PPM of free chlorine increases. This same
reading falls as the pH rises and rises as the pH drops, due
to the equilibrium between the hypochlorous acid and
hypochlorous ion concentrations. In your case, adding sodium
bicarbonate causes the pH to nudge upward, lowering the ORP
and causing the controller to add more chlorine to raise the
ORP. In order to maintain proper pH and chlorine conditions,
the pH and ORP must both be under control. So long as the pH
is within the desired 7.2-7.6 range, the controller will
keep the chlorine at the proper level. I hope that this
information proves helpful. hlorine causes the reading to rise. The
ORP is based on the hypochlorous acid concentration, which
rises as the PPM of free chlorine increases. This same
reading falls as the pH rises and rises as the pH drops, due
to the equilibrium between the hypochlorous acid and
hypochlorous ion concentrations. In your case, adding sodium
bicarbonate causes the pH to nudge upward, lowering the ORP
and causing the controller to add more chlorine to raise the
ORP. In order to maintain proper pH and chlorine conditions,
the pH and ORP must both be under control. So long as the pH
is within the desired 7.2-7.6 range, the controller will
keep the chlorine at the proper level. I hope that this
information proves helpful.
Sincerely. Alan Schuster, 8/15/2005
► Stabilizer
and Indoor Salt Chlorinator?
We have two pools/spas with chlorine
generators. I read where a stabilizer is recommended, but
these are indoor (hotel) pools, so why would I need the
cyanuric acid? Thanks for your time!
Steve, Iron River, MI, 3/37/2007
I don't know where you read that. I see no reason that
Cyanuric Acid should be required. UV is an outdoor problem.
Dichlor is used, indoors, because it is quick dissolving and
essentially pH neutral. My guess is that the instructions
were not written with an indoor pool in mind. I hope that
this information is helpful.
Sincerely. Alan Schuster, 3/27/2007
► Calculating
Bather Loads?
I have a large group coming into our
municipal pool. How can I be sure that the pool will
accommodate that number of people?
M. C., 9/8/2005
According to CPO information, the following calculations are
used.
Commercial Pools: Surface Area in Square Feet divided by 24
= maximum bathers.
Commercial Spas: Surface Area in Square Feet divided by 10 =
maximum bathers.
Please verify that this information is current or applicable
to for your area.
Sincerely. Alan Schuster, 9/9/2005
► Combined
Chlorine Issues?
This is not your standard question
regarding chloramine reduction. As a Health Inspector I deal
with this issue a lot but here I want to ask about speed.
Specifically the speed at which breakpoint can be achieved.
A health club that closes at 10:00 pm and opens at 5:00 AM
tries to beat the clock in super chlorination. With
chloramines that reach 1-2 ppm, how fast can breakpoint be
achieved with conventional shocking with liquid chlorine?
125,000 gallon lap pool. The CDC discusses contact time in
cases of liquid stool contamination. Double the dose of
chlorine results in half the time needed for contact. Can
this same principle be used in chloramine elimination? 1.0
ppm CC conventionally requires 10 ppm chlorine to reach
breakpoint (actually 7.5 or so but who's counting). What
about 1.0 ppm CC hit with 20 ppm chlorine? Potassium
peroxymonosulfate could be an option, but they do not have a
test kit to deal with the interference on testing chlorine.
Seemingly pointless, if you don't know you have reached
breakpoint. Alan, please hurry! Thanks!
Tim R., 2/16/2008
All chemical reactions, and destroying combined chlorine is
a chemical reaction, are partially governed by
concentration. This is really a physical chemistry problem,
but simply stated, time, temperature and circulation have to
be considered, as well. Raising the concentration , will cut
the reaction time, but even that is effected by variables. A
pool is full is organics, that given time, will form more
combined chlorine. If there is 1 PPM of combined chlorine at
pool closing time, adding 10 PPM of free chlorine might not
be enough, because of the organics present reacting with the
free chlorine or the formation of more combined chlorine,
from the nitrogenous wastes. Adding 10 PPM of free chlorine
could still leave you with more than 1 PPM of combined
chlorine. No two pools or situations are the same, so a set
figure might not always work. If 10-1 does not produce a
combined chlorine of less than 1 PPM, while leaving a free
chlorine level, within your state's guidelines, I can see
the utility of adding 12-1 or 15-1 or more. Potassium
monopersulfate would be an alternative worth looking into.
It might be used, in conjunction, with the regular dosing of
liquid chlorine. LaMotte Company offers
MPS test strips. I
am not sure what your state allows, in terms of alternative
sanitizing, but salt chlorine generators,
ozone generators
or UV Sterilizers can go a long way towards improving
swimming conditions and compliance with the sanitation
requirements. I hope that I have been of some help. , will cut
the reaction time, but even that is effected by variables. A
pool is full is organics, that given time, will form more
combined chlorine. If there is 1 PPM of combined chlorine at
pool closing time, adding 10 PPM of free chlorine might not
be enough, because of the organics present reacting with the
free chlorine or the formation of more combined chlorine,
from the nitrogenous wastes. Adding 10 PPM of free chlorine
could still leave you with more than 1 PPM of combined
chlorine. No two pools or situations are the same, so a set
figure might not always work. If 10-1 does not produce a
combined chlorine of less than 1 PPM, while leaving a free
chlorine level, within your state's guidelines, I can see
the utility of adding 12-1 or 15-1 or more. Potassium
monopersulfate would be an alternative worth looking into.
It might be used, in conjunction, with the regular dosing of
liquid chlorine. LaMotte Company offers
MPS test strips. I
am not sure what your state allows, in terms of alternative
sanitizing, but salt chlorine generators,
ozone generators
or UV Sterilizers can go a long way towards improving
swimming conditions and compliance with the sanitation
requirements. I hope that I have been of some help.
Sincerely. Alan Schuster, 2/16/2008
► Appearance
Of Stains?
I operate a 240,000 gallon painted
plaster outdoor pool; after using bromine for about 20 years
we switched to calcium hypochlorite in 2004. We never had
any "staining: problem using bromine, just a minor green
algae problem which we handled with the appropriate
algaecide. The summer of 2004 we experienced tan
/light brown/yellowish "staining" - on the walls and floor
of the pool, starting in the diving well, slowly moving out
into the shallower parts of the pool, gradually increasing
in quantity as the summer went on. The pool company I dealt
with said it was a metal stain and had me use metal
treatment products to take care of. Pool was shut down for
several days, chemicals added - all pertinent directions
followed, pool thoroughly brushed, backwashed sand filters
thoroughly and put pool back into operation, using a metal
treatment product on a maintenance basis. However, the
stains began to return. They can be brushed off if done
early in their appearance - but difficult considering the
size of the pool. When you brush it off water in area turns
milky. When I opened pool in 2005 since metal staining did
not seem to be the culprit we switched gears to algae
"staining." I treated pool with shock and algaecide,
initially and on a maintenance basis. But the stains
appeared same as in 2004. Only thorough brushing kept them
somewhat under control for the summer. My question is this?
How do I determine for sure what is causing the "stain" to
form, and then what to do to prevent or control it. The only
thing we changed was switching from bromine to cal-hypo.
Each spring the pool is emptied, then cleaned with TSP and
an acid wash before refilling and putting into operation. We
stay on top of everything else - free and combined chlorine,
pH, alkalinity, calcium hardness, filter cleaning, etc. We
checked the source water for the pool and nothing changed
there. The pool company I work with is at a loss. Any
ideas? Would appreciate it.
Dick W., 3/8/2016
I am not sure that is the answer, but it does fit the facts.
Bromine is acidic and will tend to keep the pH and TA
towards the lo wer end, unless adjustments are made. It does
not contribute to the calcium hardness. Minerals and metals
tend to be more soluble at lower pH ranges. Cal hypo has a
high pH and will raise the pH and TA, as product s being
added. This tends to decrease the solubility of minerals and
metals, as the pH and TA rise. Adding more and more calcium
hardness over time only makes for more problems. I suggest
that you monitor the calcium hardness and add regular doses
of scale treatment. When the calcium level exceeds 400 PPM,
scaling becomes more likely, as does metal staining. Calcium
hypo may not be the best product to use, in this pool. I
realize that there are cost considerations, but liquid
chlorine would avoid the increasing hardness levels. Have
you ever considered a salt chlorine generator? It will give
you more control and less handling and storage problems. And
the water will smell and feel better. Good luck and I hope
that I have been helpful. wer end, unless adjustments are made. It does
not contribute to the calcium hardness. Minerals and metals
tend to be more soluble at lower pH ranges. Cal hypo has a
high pH and will raise the pH and TA, as product s being
added. This tends to decrease the solubility of minerals and
metals, as the pH and TA rise. Adding more and more calcium
hardness over time only makes for more problems. I suggest
that you monitor the calcium hardness and add regular doses
of scale treatment. When the calcium level exceeds 400 PPM,
scaling becomes more likely, as does metal staining. Calcium
hypo may not be the best product to use, in this pool. I
realize that there are cost considerations, but liquid
chlorine would avoid the increasing hardness levels. Have
you ever considered a salt chlorine generator? It will give
you more control and less handling and storage problems. And
the water will smell and feel better. Good luck and I hope
that I have been helpful.
Sincerely. Alan Schuster, 3/8/2016
► Cracked
Pool Wall?
We
operate a 100,000 gallon commercial pool and recently had a
crack develop in a wall. It was repaired and painted over.
It now seems to have reopened and is probably responsible
for a significant water loss. Do you have any suggestions
for making a better repair? Thanks for any help you can
offer.
Martin M., Georgia, 4/21/2009
Most likely this is a stress crack and needs to be
stabilized. Products, such as epoxy, can be used, but don't
always work while the crack is under pressure to expand.
The Torque-Lock system is
designed to make structural concrete crack repair, that are
rock solid. If you want to make the proper repair,
this is what you want to use. Good luck and I hope that this solves the problem.
Sincerely. Alan Schuster, 4/21/2009
|
Visit The Website Stores . . . for
better informed shopping!!! |
|
 |
Shipping is FREE* . . . within
the Continental U.S.
A $9.99
handling charge will apply
to Continental U.S. Orders,
under $75.00. Orders
outside of the Continental
U.S. may require some
additional charge, based on
quantity and destination.
Most products can be
shipped World-Wide. International
and orders outside of
Continental U.S. - see
comments on the ordering
pages. |
|
 |

Major
Credit Cards and PayPal are accepted. |
|
▲
Return To Top Of Page
▲
Aqualab Systems, LLC does not
make any warranty or
representation, either expressed
or implied, regarding the
accuracy or completeness of the
information provided by this
website; nor does Aqualab
Systems., LLC. assume any
liability of any kind whatsoever
related to, or resulting from,
any use or reliance on this
information. The content of this
website should not be used, if
it is conflict with any
applicable federal, state or
local regulations or guidelines.
© Aqualab Systems, LLC. All
rights reserved
|
|



























